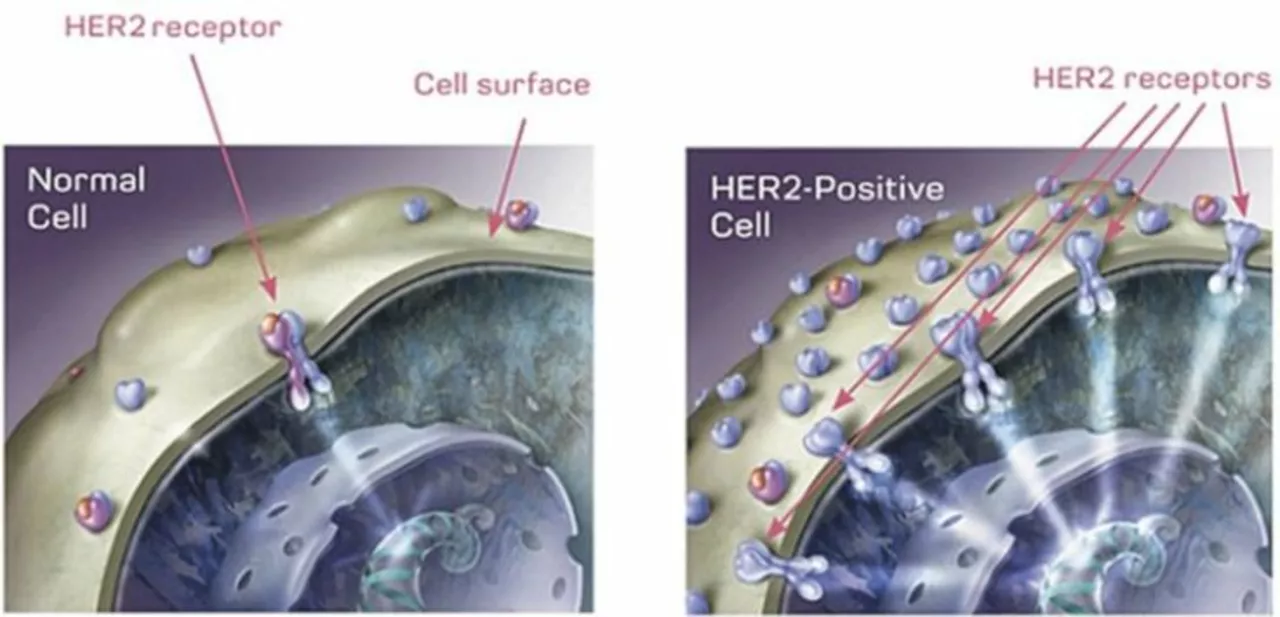
Understanding HER2-Positive Breast Cancer
Before diving into the potential of alpelisib in the treatment of HER2-positive breast cancer, it is crucial to understand what exactly HER2-positive breast cancer is. HER2 stands for human epidermal growth factor receptor 2, which is a protein found on the surface of breast cells. In some cases, breast cells can produce too much HER2, leading to the rapid growth and division of cancer cells. This type of breast cancer is known as HER2-positive breast cancer, and it affects approximately 20% of breast cancer patients.
HER2-positive breast cancer can be aggressive and tends to grow faster than other types of breast cancer. However, recent advancements in targeted therapies have improved the prognosis for patients with this type of cancer. One such promising targeted therapy is alpelisib, a drug that has shown potential in the treatment of HER2-positive breast cancer.
Alpelisib: A New Targeted Therapy
Alpelisib is a type of targeted therapy known as a PI3K inhibitor. The PI3K pathway is a cellular signaling pathway that plays a crucial role in cell growth, proliferation, and survival. It is often overactivated in cancer cells, leading to uncontrolled growth and division. By inhibiting the PI3K pathway, alpelisib can slow down the growth of cancer cells and, in some cases, even cause them to die.
What makes alpelisib unique is that it specifically targets the PI3K alpha isoform, which is the most frequently mutated form of PI3K in breast cancer. This selectivity means that alpelisib may be more effective and have fewer side effects than other PI3K inhibitors.
Early Clinical Trials and Promising Results
Initial clinical trials have shown promising results for the use of alpelisib in the treatment of HER2-positive breast cancer. In a Phase I study, patients with advanced HER2-positive breast cancer who had previously been treated with trastuzumab and pertuzumab were given alpelisib in combination with these targeted therapies. The results showed that the combination of alpelisib, trastuzumab, and pertuzumab was well-tolerated and demonstrated antitumor activity.
Another study, known as the SOLAR-1 trial, investigated the use of alpelisib in combination with fulvestrant in patients with hormone receptor-positive, HER2-negative advanced breast cancer. The results of this trial showed that the combination of alpelisib and fulvestrant significantly improved progression-free survival compared to fulvestrant alone.
Addressing Treatment Resistance
One of the major challenges in treating HER2-positive breast cancer is the development of resistance to targeted therapies. Over time, cancer cells can develop mutations that allow them to evade the effects of these drugs, leading to treatment resistance. The PI3K pathway is one of the key pathways implicated in the development of resistance to HER2-targeted therapies.
Alpelisib's ability to target the PI3K pathway may provide a new approach to overcoming treatment resistance in HER2-positive breast cancer. By inhibiting the PI3K pathway, alpelisib may help to resensitize cancer cells to HER2-targeted therapies, ultimately improving treatment outcomes.
Future Directions: Combination Therapies
As with many targeted therapies, the future of alpelisib in the treatment of HER2-positive breast cancer may lie in combination therapies. By combining alpelisib with other targeted therapies, such as trastuzumab and pertuzumab, researchers hope to increase the effectiveness of treatment and overcome treatment resistance.
Further clinical trials are needed to determine the most effective combinations and dosing strategies for alpelisib in the treatment of HER2-positive breast cancer. Additionally, researchers are investigating the use of alpelisib in other types of breast cancer, as well as in other types of cancer that may have mutations in the PI3K pathway.
Addressing Side Effects and Safety
As with any cancer treatment, the potential side effects and safety of alpelisib must be considered. Common side effects of alpelisib include diarrhea, nausea, rash, and fatigue. In the SOLAR-1 trial, the most common severe side effect was high blood sugar, which occurred in approximately 37% of patients receiving alpelisib.
While these side effects can be managed with dose adjustments and supportive care, it is essential for researchers to continue to monitor the safety and tolerability of alpelisib in combination with other targeted therapies.
Conclusion: A Promising Future for Alpelisib
In conclusion, alpelisib holds significant potential for the treatment of HER2-positive breast cancer. Its unique ability to target the PI3K pathway, which is often implicated in treatment resistance, may provide a new approach to overcoming this challenge. Furthermore, its use in combination with other targeted therapies may lead to more effective treatment options for patients with this aggressive form of breast cancer.
As research continues and more clinical trials are conducted, we can hope that alpelisib will become an important tool in the arsenal of targeted therapies for HER2-positive breast cancer, ultimately improving treatment outcomes and the quality of life for patients.

jalyssa chea
May 31, 2023 AT 03:54also why is everyone acting like this is a miracle when the side effects are wild like high blood sugar??
Gary Lam
June 2, 2023 AT 02:54Also, alpelisib? More like alpelisib-why-did-i-agree-to-this.
Peter Stephen .O
June 3, 2023 AT 04:54most PI3K inhibitors are like throwing a grenade in a room - everything gets hit
but alpelisib? It’s the quiet guy who walks in, picks the right target, and leaves the rest untouched
and yeah the hyperglycemia is rough but hey - we’ve got metformin for that
imagine if we could pair this with CDK4/6 inhibitors next - that’s the real dream team
we’re not just treating cancer anymore we’re hacking the cell’s OS
Andrew Cairney
June 3, 2023 AT 13:19you think they’re really trying to help patients or just testing how fast they can get FDA approval before the next quarterly earnings call?
37% get high blood sugar? That’s not a side effect - that’s a feature they didn’t tell you about
and don’t get me started on the ‘combination therapy’ hype - they’re just layering drugs to make the trial stats look better
you ever wonder why all these ‘breakthroughs’ come from the same 3 pharma companies?
they’re not curing cancer - they’re creating lifelong customers
:(
Rob Goldstein
June 4, 2023 AT 14:05the SOLAR-1 data was solid but we need to contextualize it - PFS improvement was ~11 months vs 5.7, which is huge in metastatic disease
the real win is in overcoming resistance to trastuzumab/pertuzumab - PI3K/AKT/mTOR is the classic escape route
yes, hyperglycemia is common but manageable with metformin or SGLT2 inhibitors
and the combo potential with CDK4/6i or mTORi? That’s where the next phase trials are headed
we’re not just extending survival - we’re buying time for immunotherapy to catch up
if you’re a clinician, this is one of the most actionable targets we’ve had in years